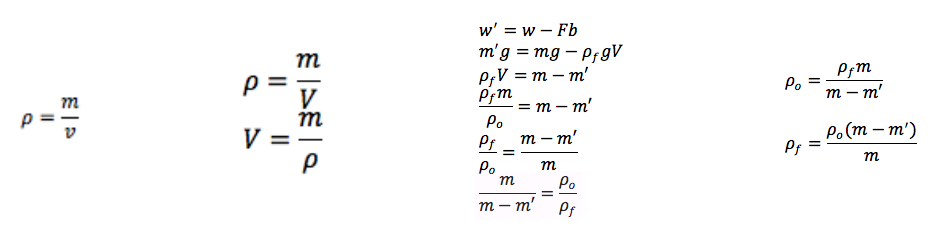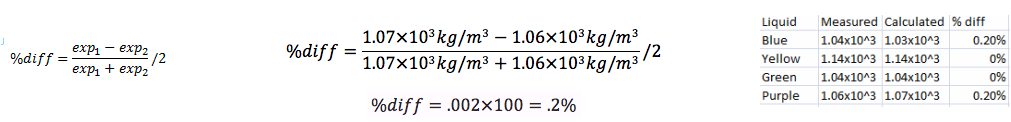Density of Different liquids
Lab Partners: Kyle Troch, Michael Perkins & Ben Ronemus
Date Completed: May 22, 2014
Date Completed: May 22, 2014
Purpose
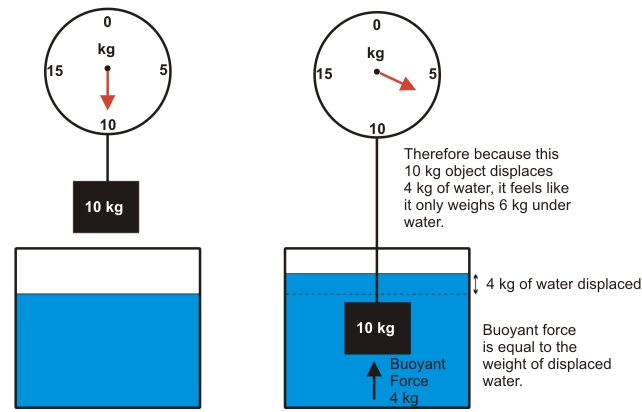
The purpose of this lab is to use Archimedes' Principle to fine the density of different solutions of water and salt using a know mass of a cube of brass.
Theory
Archimedes' Principle states that buoyant force on a body immersed in a fluid is equal to the weight of the fluid displaced by that object. In our equations we used m' to stand for apparent mass. Apparent mass is what an object appears to weigh in a liquid because liquids have a buoyant force that counteracts gravity. This would make the mass appear to be less even though it has the same mass. In addition to using equations from Archimedes' Principle we used the density equation.
Experimental Technique
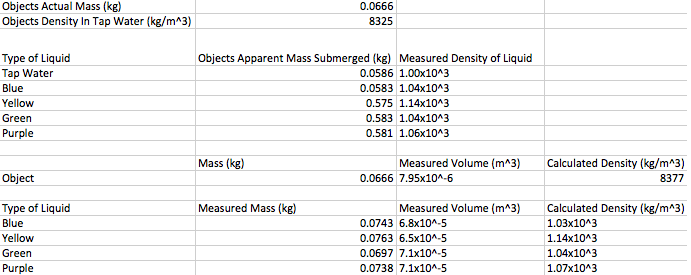
We measured the mass and apparent mass of the cube by using a hanging device attached to a balance. Then to find apparent mass however we had to submerge the object in one of the liquids to measure it. After we found the two different masses we could put those into our equation to find the density of each liquid. Then we poured the liquids into individual graduated cylinders to measure the volume and mass of the liquids. Once we knew those two measurements we could calculate the liquids density using the density equation that way we could compare our measured density to the actual density.
Green Liquid Yellow Liquid
Blue Liquid Purple Liquid
Data & Analysis
Conclusion
The purpose of this lab was to use Archimedes' Principle to find the densities of different liquids. After completing the lab we found out that it was very successful. We were able to measure and calculate all of the densities of all of the liquids, and then compare them to find out that they are all very very close. This makes me believe that our lab was very successful. Some of the things that could cause error were air bubbles in the fluids, evaporation, and the meniscus in the graduated cylinder. Even though there are those possibilities of error we kept those to a minimum while completing this lab, and it shows in our results i believe. The reason I am so confident in our results is because the green and yellow liquids had a percent difference of 0, and the blue and purple liquids both had .2% for their percent difference. All in all i believe this lab was very successful and we obtained very accurate answers.
References
http://physics.weber.edu/carroll/archimedes/principle.htm
References
http://physics.weber.edu/carroll/archimedes/principle.htm